The management of perioperative nutrition in patients with end stage liver disease undergoing liver transplantation
Prevalence
There is an association between nutritional status and postoperative mortality (1,2). Malnutrition is a risk in form of deficiencies of energy, protein and nutrients, which influences some organs’ function and leads to bad clinical outcomes regardless of etiology. Malnutrition is common in patients with liver cirrhosis especially end stage liver disease (ESLD). And still, there is a percentage as much as 48% existing insufficient calories in patients even with liver Child-Pugh grade A score. Severity of malnutrition correlates closely with complications of the decompensated liver disease and is an independent risk factor to predict the clinical outcome of this population (3-5). Reason for malnutrition in patients with ESLD is multifactorial. However, major determinants are decreased nutrient and caloric intake, intestinal malabsorption, and overloaded catabolism.
Inadequate nutrients intake was seen under a variety of reasons, such as loss of appetite caused by tristimania, anorexia, drug side effects and satiety caused by less gastrointestinal peristalsis and gastric restrictive expansion caused by large volume ascites. In addition, acute gastroesophageal varicose hemorrhage followed by long time fasting is also common in clinical practice. Professor Plauth pointed out in a research that many clinicians make a prescription of low protein diet to avoid encephalopathy, which leads to deterioration of nutritional status. However, it is not necessary and lack of being evidence-based in most cases (6).
One other important factor is the presence of impaired absorption function due to portal hypertension (7), which frequently accompanied with portal hypertensive gastrointestinal disease or peptic ulcer disease. Under this condition, there is gradually arising impaired absorption of fat-soluble vitamins such as A, D, E and K, intestinal mucosal atrophy and conditional bacterial infections, followed by hypercatabolic status and higher mortality. Finally, iatrogenic factors such as the multiple hospitalizations, pending examinations and procedures (e.g., paracentesis) should not be ignored as well.
Hypermetabolic status
The liver as the largest metabolic organ plays a central role in regulating energy, protein, and lipid metabolism and integrating a wide variety of complex biochemical process including excretion of endogenous and exogenous useful hormones. Severe liver injury could result in significant metabolic derangement, especially in patients with ESLD who are characterized by abnormalities of hypermetabolic status.
Liver transplantation (LT) candidates with ESLD often in the setting of hyperdynamic circulation present disturbances in body composition and in the hypermetabolic rate. Energy expenditure is determined by the measurement of the basal energy expenditure (BEE), which can be calculated using the Harris-Benedict equations. There is up to 34% in cirrhotic patients, who are considered hypermetabolic with resting energy expenditure (REE) 120% of the expected (8). Fever, spontaneous peritonitis and bacterial translocation are undoubtedly considered as the most common inducing factors contributing to accelerating catabolism.
Metabolism abnormity of the three nutritional substances
The prevalence of glucose intolerance and insulin resistance is seen in the population of ESLD patients. Many could develop the hepatic diabetes. Overnight fasting, hepatic glycogen stores are depleted in patients with ESLD, presenting increased gluconeogenesis from amino acids and increased lipid peroxidation. At this time, fat becomes their main substrate for energy. As the result, the mobilization of amino acids from the skeletal muscles and visceral proteins is active, demonstrating muscle depletion and decrease in subcutaneous fat.
Perioperative maintenance of normoglycemia is frequently emphasized to improve surgical outcomes and may be quite important factor to prevent surgical site infection (9). A research found that surgical site infections were reduced in patients whose HbA1c was <7% (10). In additionally, hyperglycemia has significant influence on gastric and small bowel motility. It seems that there is a linear progression of decreased motility and glucose level when the glucose level goes over 200 mg/dL (11).
Patients with ESLD have impaired synthesis of polyunsaturated fatty acids and essential fatty acids because of impaired liver function (12). Decreased polyunsaturated fatty acids have been associated with the severity of malnutrition in liver disease (13). As mentioned above, cholestasis and portal hypertensive enteropathy may cause impaired absorption of fat and fat-soluble vitamins. This can result in specific deficiencies such as vitamin A deficiency, osteoporosis because of vitamin D loss (14), and coagulopathy due to vitamin K loss. Decreased levels of folate, B12, zinc, magnesium, calcium and phosphorus are also common.
Loss of protein and trace elements is a common clinical phenomenon in ESLD patients resulting from complications of cirrhosis or iatrogenic interventions. Patients with ESLD have a protruding clinical manifestation of intractable encephalopathy, which accompanies with laboratory test of imbalance of branched-chain amino acids (BCAA) and aromatic amino acids. High level of aromatic acids promote cerebral tissue synthesize more harmful neurotransmitters to compete with endogenous neurotransmitters which in return make further damage to the brain cells. In view of this, many physicians prescribe a diet of restrictive protein leading to a common iatrogenic factor. Some other common iatrogenic interventions are the use of diuretics in order to cope with ascites and fluid retention, the use of lactulose, and the performance of repeated paracentesis.
Nutrition support for patients with chronic ESLD prior to LT
As elaborated above, patients with ESLD have an altered metabolic response to starvation and overnight fasting can result in muscle depletion. Daytime or evening snacks are therefore important. Clinicians should consider in advance nutritional problems when admitting patients with cirrhosis and try their best to provide supplementation in time and meet the nutritional needs of these patients as soon as possible. Early intervention in replenishing the nutrient deficit can prolong life expectancy, ameliorate quality of life, diminish complications and prepare them for a more successful LT (15).
There is definite association between nutritional state and surgical outcomes in many studies (16-19). These research outcomes reveal significant increased morbidity and other clinical indices such as increased infection complications (e.g., surgical site infections), increased intensive care unit (ICU) admissions and shorter length of stay (LOS). In a study (20), the complication rate was significantly lower in patients accepting preoperative nutrition (parenteral or enteral) with a high risk nutrition state for at least 7 days compared with the control group without preoperative nutrition support (25.6% vs. 50.6%, P=0.008). The postoperative hospital stay was significantly shorter in the preoperative nutrition group. However, the complication rate and the postoperative hospital stay were similar between patients in slight-moderate risk nutrition state with and those without preoperative nutritional support. This finding suggests that adequate preoperative nutritional support (≥7 d) is beneficial to patients with a nutrition risk score (NRS) score at least 5.
It is worth attention, although numerous studies have demonstrated conclusively value of preoperative nutritional support in patients undergoing surgery, few studies have been published concerning preoperative nutritional support in hepatic surgery in comparison with the lots of studies published in other fields in recent 20 years, not to mention of the LT. And still, there are many unresolved issues and controversies in the field of LT. First, there is lack of adequate cases and randomized cohort trials, failing to achieve a positive conclusion in the relationship between preoperative nutritional status and post LT outcome particularly mortality. Second, no widely agreed standard of nutrition assessment was provided to clinicians, which makes it difficult to interpret which patient truly needs preoperative nutrition support in settings of different population, route of nutrition delivery and different nutrition formula. Recent two prospective studies conducted on high risk or severe patients indicated that more benefit be in patients with more higher risk or severe patients (20,21). Finally, lots of other crucial factors play an important role in influencing post LT mortality other than perioperative nutrition intervention. Some researchers also failed to yield a significant decrease in mortality after LT by means of nutrition intervention preoperatively (22-26). The confusions derived from heterogeneity in the above studies become partly an answer to a question “why is perioperative nutrition support widely accepted, but not widely practiced?” (27).
Nutritional assessment
An overall perioperative assessment usually includes a history, physical examination, laboratory test and instrumental examination focusing on risk factors for cardiac, pulmonary, and infectious complications, along with a determination of a patient’s functional capacity. To our disappointment, conventional parameters usually used for the nutritional assessment are one-sided and no gold standard could cover the extent of malnutrition during the perioperative stage in population of patients with ESLD awaiting LT (28). It suggests that comprehensive assessment should be better.
Clinicians should pay an attention to a complete dietary history and present diet condition when admitting patients with ESLD. Representative symptom indices include weight loss in the recent 6 months, anorexia degree, declined appetite, feeling of fullness, nausea or vomiting, diarrhea and constipation. The ability of oral intake is connected with postoperative mortality. In addition, body mass index (BMI) should be obtained from patient’s height and weight. BMI of <18.5 kg/m2 or the 15th percentile for arm anthropometric measurements often were considered be malnutrition in this population. However, BMI can be inaccurate in the condition of fluid retention or edema. Besides a complete history, other assessment tools have been applied in clinical practice.
Subjective global assessment (SGA) first brought out by Detsky in 1987 (29), which was recommended as a simple bedside method by European Society for Parenteral and Enteral Nutrition and Metabolism (ESPEN) (30). This nutritional evaluation tool is based on weight changes, dietary changes, gastrointestinal symptoms (including nausea, vomiting, diarrhea or constipation), functional capacity, stress of the disease and muscle wasting (Table 1). The advantage of this tool lies in simplification, good reproducibility and non-invasive laboratory test. It can significantly correlate with body composition in the condition of severe malnutrition and will be a prognostic predictor in complications after LT (36). SGA, anthropometric measurements and the functional index of handgrip strength are more commonly used in nutritional assessment. It’s limitation appears to be excessive subjectivity, low sensitivity (37) and failed to distinguish patients with mild malnutrition. It speaks more to patients with chronic or predefined nutritional deficiency and cannot reflect acute phase of nutritional changes (31). The succeeding modified SGA-Royal Free Hospital-Subjective Global Assessment (RFH-SGA) (38), which combines both subjective and objective parameters including measures of BMI calculated from dry weight, mid-arm muscle circumference (MAMC), and dietary intake could be more promising in nutritional assessment prior to LT. In a study (39), Monsef et al. compared the agreement of different tools including SGA, RFH-SGA, anthropometry and biochemical tests to assess the nutritional statues in patient with ESLD prior to LT. Agreement among all the methods, RFH-SGA showed perfect agreement with SGA as performed by the kappa test (K=0.81). Also, RFH-SGA is an independent factor associated with mortality in pretransplant patents from multivariable models (15).
NRS 2002 was based on 128 randomized trials. In each trial, and the group of patients was classified with respect to nutritional status and severity of disease. The screening system appeared to be able to distinguish between trials with a positive effect or no effect, and it can therefore probably identify patients who are likely to benefit from nutritional support (32). One limitation of this protocol is that weight loss is inaccurate if patients cannot be weighed due to unconsciousness, or body weight is unreliable due to fluid accumulation. However, it is useful to translate the BMI values to measurements of mid-arm circumference (MAC) (40). Decades of RCTs suggested that a MAC <25 cm corresponds to a BMI <20.5. But, these data did not allow for distinguishing between values for MAC corresponding to a BMI <18.5 vs. 18.5-20.5. Another weakness is that it cannot provide individualized nutritional proposal (nutritional requirement, ways, gastrointestinal tolerance and the transitional scheme of nutritional support, etc.) and identified by only grade E evidence supporting the guideline to screen hospitalized patients for nutritional risk by American Society for Parenteral and Enteral Nutrition (ASPEN) guidelines (41). However, the system has been widely introduced in hospital. Nurses and doctors can use the tool after a short time training to make nutritional plans as soon as possible according to this system scores for severity of the disease. In addition, the present screening system is now recommended by ESPEN for nutritional screening in hospitals together with the malnutrition universal screening tool (MUST) system in the community and the mini nutritional assessment (MNA) system in institutionalized elderly population (Table 1).
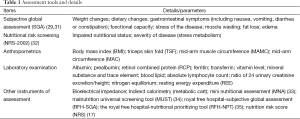
Full table
Some study attended to obtain a relationship between some common nutritional evaluation system by Child-Pugh classification and postoperative outcomes (24). Additional literatures can be available other than studies about nutrition assessment in the field of LT. For example, Schiesser et al. compared the prognostic efficacy of 3 score systems in incidence and severity of postoperative complications in patients undergoing GI surgery. It was found that only nutrition risk score (NRS) and malignancy remained prognostic factors for the development of complications with odds ratios of 4.2 (P=0.024) and 5.6 (P<0.001), respectively. The NRS was the best score in predicting patients who will develop complications in this population (17). The correlation between the other two systems (nutrition risk index and bioelectrical impedance) and NRS was weak.
Administration after LT
How to determine the nutritional requirement and target patients in post liver transplant population is far from easy. Surgery has been associated with hypermetabolism in reported literatures and guidelines (42,43). During the stay of surgical intensive care unit (SICU), nutritional support should be emphasized on the destination of graft function recovery and overall convalescence which is faced with stress from critical illness and multiple treatments (mechanical ventilation, hemodiafiltration, use of corticosteroid and immunosuppressive agents and so on).
Several methods are usually used to predict patient’s nutritional requirement, such as mathematical formula of Harris Benedict equation and indirect calorimetry. Accurate determination of REE is necessary to ensure the energy needs are met and to avoid the postoperative complications and in the SICU setting. The formula of 25 kcal/kg ideal body weight provides an approximate estimate of daily energy requirements. Under conditions of severe stress, requirements may approach 30 kcal/kg ideal body weight and a protein intake of 1.5 g/kg ideal body weight (or approximately 20% of total energy requirements) is generally effective to limit nitrogen losses (supported based on grade B evidence) (42). Some studies suggest that the degree of hypermetabolism is on average not more than 110-120% of predicted (44,45), and should not be furnished more than 20-25 kcal/kg daily in the acute phase of critical severe illness (46).
In general, the total amount of calories should be provided at least 1.2 times the BEE and a mixed-fuel system of both carbohydrate and fat is suggested to provide energy in post-OLT period. Usually, 70% of non-protein calories are given as carbohydrates during this phase (47). In a study (48) of patients in a surgical critical care unit, either 30 kcal/kg adjusted body weight or the REE calculated from the Harris Benedict equation multiplied by 1.5 adequately predicts the nutritional requirements of critically ill surgery. In some other settings of occurrence of encephalopathy (49) and acute kidney failure performing routing hemodialysis (50,51) post LT, there are some different energy requirements according to weight, age, gender and physical activity.
Appropriate and timely nutrition intervention becomes more and more recognized in agreement. In regard to the starting time of nutrition, early postoperative enteral nutrition support is beneficial in decreasing mortality and morbidity not only in the field of gastrointestinal surgery (52,53) but also of LT (54) and suggested in some guidelines (46,55). Marik et al. (56) carried out a systematic review on the effect of early enteral nutrition (defined as the initiation of enteral feeding within 36 h of admission or within 36 h after surgery) on the outcome of critically ill and injured patients (15 studies including 753 patients), which demonstrated the benefit of the early initiation of enteral nutrition in lower incidence of infections. But there were no significant differences in mortality or noninfectious complications between the two groups.
Additional several meta-analysis show more clearly the evidence of feeding started within 24-48 h of surgery. Recent clinical trial (57) performed in 346 liver transplant patients has gotten a conclusion that Bacterial sepsis occurred in 5.9% of patients who received tube feeding within 48 hours of surgery compared with 21% in patients who started tube feeding after 48 hours. Otherwise, for those patients in hemodynamic instability requiring vasopressor agents (58,59), once the hemodynamic is stable for more than 24 hours and bowl function is in recovery, feeding should commence from at 10-20 mL/h rate with a progressive increase to reach the full goal within 2-3 days of post-operation exception of contraindication for enteral nutrition (43).
Some other advantages of enteral nutrition were reflected in its cost-effective (60) and maintaining the integrity of gut. Mehta et al. (61) compared the effectiveness of enteral nutrition through jejunostomy tube (TF) placed at the time of LT for immediate postoperative nutrition support to total parenteral nutrition (TPN). Achieving adequate oral intake occurred more quickly in TF group (median, 19.5 days) than in TPN group (median, 38.6 days). And also, a lower frequency of postoperative ileus was in the TF group than that in the TPN group (8.3% vs. 33.3%, respectively). The S.C.C.M and A.S.P.E.N guidelines (55) in 2009 pointed out that if the patient is malnourished preoperatively, surgery should be delayed to initiate parenteral nutrition for 7 days preoperatively, and then continued for at least 7 days in the postoperative period (grade B evidentiary support). PN should only be used when EN is not possible. If the patient is well nourished before the surgery, one should delay parenteral nutrition for 5-7 days other than initiating immediately (only when EN is not available).
However, there was still some controversy of enteric versus parenteral nutrition in perioperative nutritional support applied to patients undergoing LT. A randomized trial in 24 patients, comparing enteral feeding via a nasojejunal tube and TPN after LT, did not show any significant difference in the median days to start eating, nutritional parameters, early postoperative absorptive capacity and intestinal permeability (62). Similar results are in a meta-analysis study (63) emphasizing the misplaced enthusiasm that early enteral nutrition as compared with early parenteral nutrition would reduce mortality.
Specialized nutritional agents have been shown to improve outcome in specific critically ill patients including post LT. It appears that each agent may have different effects on patient outcome, making it difficult to make consistent and definite conclusion due to disparity of literatures. Related literatures about impact on outcome of the post liver transplant population are listed in the Table 2. The literatures showed a trend to decrease overall bacterial infections and morbidity, although they are small-group studies in large part.
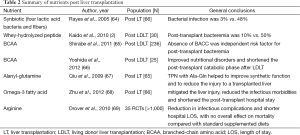
Full table
Some studies (2,70) evaluated the effects of enteral immune-modulating diet on postoperative infectious complications finding out that much less incidence of infection in patients who received immunonutrition. Likewise, Kaido et al. in 2012 retrospectively analyzed the advantage of immunomodulating diet containing hydrolyzed whey peptide in post living donor LT patients (76 consecutive adult patients) based on the literature of 2 years ago (71). Shirabe et al. (65) retrospectively stated that BCAA supplementation could reduce the incidence of bacteremia (BCAA supplementation: 6.7% vs. control: 22.0%) after living donor LT. However, the ambiguity in the literature goes back for more than 20 years. A randomized trial (72) compared postoperative TPN using standard amino acids or relatively more BACC with no nutritional supplementation support in 28 patients after LT. Therapy was continued for 7 days postoperatively. Both groups with nutritional supplementation had significantly less negative nitrogen balance, shorter ICU stay and lower total hospital charges, but there was no significant difference between the two TPN groups in above clinical indices and amino acids level. It concluded that there was no additional benefit from supplemental BCAA.
The evidence of Supplementation with ω-3 fatty acids prohibiting inflammatory effect, modulating protein synthesis, and significantly ameliorating the necrotized liver injury can be found in the literature (68). In addition, overwhelming RCTs’ data showing significant benefit from formula containing arginine has been reported in the study (69).
Prospect and summary
LT has been established as a viable option for patients with end-stage liver disease. The research on transplant nutrition is making more and more progress in parallel with the surgical techniques, although there are still some ambiguities and controversies. The primary nutritional goal in the perioperative period is to provide replenishment of nutrient depleted stores, perform certain functional effect and promote overall recovery from this major surgery. It is necessary in advance to realize the importance of nutrition on perioperative patients with ESLD undergoing liver transplant patients including pre- and post-surgery period rather than providing nutrition just as a caloric source regardless of the pathological and physiological state.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Kaido T, Egawa H, Tsuji H, et al. In-hospital mortality in adult recipients of living donor liver transplantation: experience of 576 consecutive cases at a single center. Liver Transpl 2009;15:1420-5. [PubMed]
- Kaido T, Mori A, Ogura Y, et al. Impact of enteral nutrition using a new immuno-modulating diet after liver transplantation. Hepatogastroenterology 2010;57:1522-5. [PubMed]
- Hasse JM. Nutrition assessment and support of organ transplant recipients. JPEN J Parenter Enteral Nutr 2001;25:120-31. [PubMed]
- Alberino F, Gatta A, Amodio P, et al. Nutrition and survival in patients with liver cirrhosis. Nutrition 2001;17:445-50. [PubMed]
- Buyse S, Durand F, Joly F. Nutritional assessment in cirrhosis. Gastroenterol Clin Biol 2008;32:265-73. [PubMed]
- Plauth M, Schütz ET. Cachexia in liver cirrhosis. Int J Cardiol 2002;85:83-7. [PubMed]
- Tsiaousi ET, Hatzitolios AI, Trygonis SK, et al. Malnutrition in end stage liver disease: recommendations and nutritional support. J Gastroenterol Hepatol 2008;23:527-33. [PubMed]
- Müller MJ, Böttcher J, Selberg O, et al. Hypermetabolism in clinically stable patients with liver cirrhosis. Am J Clin Nutr 1999;69:1194-201. [PubMed]
- Ata A, Lee J, Bestle SL, et al. Postoperative hyperglycemia and surgical site infection in general surgery patients. Arch Surg 2010;145:858-64. [PubMed]
- Dronge AS, Perkal MF, Kancir S, et al. Long-term glycemic control and postoperative infectious complications. Arch Surg 2006;141:375-80; discussion 380. [PubMed]
- Bulat R. Delayed gastric emptying and glucose control in early type 2 diabetes. J Diabetes Complications 2013;27:4-5. [PubMed]
- Cabré E, Gassull MA. Nutritional and metabolic issues in cirrhosis and liver transplantation. Curr Opin Clin Nutr Metab Care 2000;3:345-54. [PubMed]
- Cabré E, Abad-Lacruz A, Núñez MC, et al. The relationship of plasma polyunsaturated fatty acid deficiency with survival in advanced liver cirrhosis: multivariate analysis. Am J Gastroenterol 1993;88:718-22. [PubMed]
- Gundling F, Teich N, Strebel HM, et al. Nutrition in liver cirrhosis. Med Klin (Munich) 2007;102:435-44. [PubMed]
- Gunsar F, Raimondo ML, Jones S, et al. Nutritional status and prognosis in cirrhotic patients. Aliment Pharmacol Ther 2006;24:563-72. [PubMed]
- Awad S, Lobo DN. Metabolic conditioning to attenuate the adverse effects of perioperative fasting and improve patient outcomes. Curr Opin Clin Nutr Metab Care 2012;15:194-200. [PubMed]
- Schiesser M, Kirchhoff P, Müller MK, et al. The correlation of nutrition risk index, nutrition risk score, and bioimpedance analysis with postoperative complications in patients undergoing gastrointestinal surgery. Surgery 2009;145:519-26. [PubMed]
- Marik PE, Zaloga GP. Immunonutrition in high-risk surgical patients: a systematic review and analysis of the literature. JPEN J Parenter Enteral Nutr 2010;34:378-86. [PubMed]
- Harrison J, McKiernan J, Neuberger JM. A prospective study on the effect of recipient nutritional status on outcome in liver transplantation. Transpl Int 1997;10:369-74. [PubMed]
- Jie B, Jiang ZM, Nolan MT, et al. Impact of preoperative nutritional support on clinical outcome in abdominal surgical patients at nutritional risk. Nutrition 2012;28:1022-7. [PubMed]
- Khalid I, Doshi P, DiGiovine B. Early enteral nutrition and outcomes of critically ill patients treated with vasopressors and mechanical ventilation. Am J Crit Care 2010;19:261-8. [PubMed]
- Le Cornu KA, McKiernan FJ, Kapadia SA, et al. A prospective randomized study of preoperative nutritional supplementation in patients awaiting elective orthotopic liver transplantation. Transplantation 2000;69:1364-9. [PubMed]
- Figueiredo F, Dickson ER, Pasha T, et al. Impact of nutritional status on outcomes after liver transplantation. Transplantation 2000;70:1347-52. [PubMed]
- Abbott WJ, Thomson A, Steadman C, et al. Child-Pugh class, nutritional indicators and early liver transplant outcomes. Hepatogastroenterology 2001;48:823-7. [PubMed]
- de Luis DA, Izaola O, Velicia MC, et al. Impact of dietary intake and nutritional status on outcomes after liver transplantation. Rev Esp Enferm Dig 2006;98:6-13. [PubMed]
- Merli M, Giusto M, Gentili F, et al. Nutritional status: its influence on the outcome of patients undergoing liver transplantation. Liver Int 2010;30:208-14. [PubMed]
- Martindale RG, McClave SA, Taylor B, et al. Perioperative nutrition: what is the current landscape? JPEN J Parenter Enteral Nutr 2013;37:5S-20S. [PubMed]
- Campos AC, Matias JE, Coelho JC. Nutritional aspects of liver transplantation. Curr Opin Clin Nutr Metab Care 2002;5:297-307. [PubMed]
- Detsky AS, McLaughlin JR, Baker JP, et al. What is subjective global assessment of nutritional status? JPEN J Parenter Enteral Nutr 1987;11:8-13. [PubMed]
- Plauth M, Cabré E, Campillo B, et al. ESPEN Guidelines on Parenteral Nutrition: hepatology. Clin Nutr 2009;28:436-44. [PubMed]
- Sacks GS, Dearman K, Replogle WH, et al. Use of subjective global assessment to identify nutrition-associated complications and death in geriatric long-term care facility residents. J Am Coll Nutr 2000;19:570-7. [PubMed]
- Kondrup J, Rasmussen HH, Hamberg O, et al. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr 2003;22:321-36. [PubMed]
- Guigoz Y, Lauque S, Vellas BJ. Identifying the elderly at risk for malnutrition. The Mini Nutritional Assessment. Clin Geriatr Med 2002;18:737-57. [PubMed]
- Stratton RJ, King CL, Stroud MA, et al. ‘Malnutrition Universal Screening Tool’ predicts mortality and length of hospital stay in acutely ill elderly. Br J Nutr 2006;95:325-30. [PubMed]
- Amodio P, Bemeur C, Butterworth R, et al. The nutritional management of hepatic encephalopathy in patients with cirrhosis: International Society for Hepatic Encephalopathy and Nitrogen Metabolism Consensus. Hepatology 2013;58:325-36. [PubMed]
- Campos AC, Matias JE, Coelho JC. Nutritional aspects of liver transplantation. Curr Opin Clin Nutr Metab Care 2002;5:297-307. [PubMed]
- Sanchez AJ, Aranda-Michel J. Nutrition for the liver transplant patient. Liver Transpl 2006;12:1310-6. [PubMed]
- Morgan MY, Madden AM, Soulsby CT, et al. Derivation and validation of a new global method for assessing nutritional status in patients with cirrhosis. Hepatology 2006;44:823-35. [PubMed]
- Monsef WA, Mostafa I, Zaky D. Assessment of the Nutritional Status of the Egyptian Patient with End Stage Liver Disease Prior to Liver Transplantation. Open Journal of Gastroenterology 2014;4:159-69.
- Kondrup J, Allison SP, Elia M, et al. ESPEN guidelines for nutrition screening 2002. Clin Nutr 2003;22:415-21. [PubMed]
- Mueller C, Compher C, Ellen DM, et al. A.S.P.E.N. clinical guidelines: Nutrition screening, assessment, and intervention in adults. JPEN J Parenter Enteral Nutr 2011;35:16-24. [PubMed]
- Braga M, Ljungqvist O, Soeters P, et al. ESPEN Guidelines on Parenteral Nutrition: surgery. Clin Nutr 2009;28:378-86. [PubMed]
- Singer P, Berger MM, Van den Berghe G, et al. ESPEN Guidelines on Parenteral Nutrition: intensive care. Clin Nutr 2009;28:387-400. [PubMed]
- Reid CL. Nutritional requirements of surgical and critically-ill patients: do we really know what they need? Proc Nutr Soc 2004;63:467-72. [PubMed]
- Zauner A, Schneeweiss B, Kneidinger N, et al. Weight-adjusted resting energy expenditure is not constant in critically ill patients. Intensive Care Med 2006;32:428-34. [PubMed]
- Weimann A, Braga M, Harsanyi L, et al. ESPEN Guidelines on Enteral Nutrition: Surgery including organ transplantation. Clin Nutr 2006;25:224-44. [PubMed]
- Campos AC, Matias JE, Coelho JC. Nutritional aspects of liver transplantation. Curr Opin Clin Nutr Metab Care 2002;5:297-307. [PubMed]
- Davis KA, Kinn T, Esposito TJ, et al. Nutritional gain versus financial gain: The role of metabolic carts in the surgical ICU. J Trauma 2006;61:1436-40. [PubMed]
- Kim SJ, Kang YG, Park JH, et al. Effects of low-intensity laser therapy on periodontal tissue remodeling during relapse and retention of orthodontically moved teeth. Lasers Med Sci 2013;28:325-33. [PubMed]
- Toigo G, Aparicio M, Attman PO, et al. Expert working group report on nutrition in adult patients with renal insufficiency (Part 2 of 2). Clin Nutr 2000;19:281-91. [PubMed]
- Cano NJ, Aparicio M, Brunori G, et al. ESPEN Guidelines on Parenteral Nutrition: adult renal failure. Clin Nutr 2009;28:401-14. [PubMed]
- Lewis SJ, Andersen HK, Thomas S. Early enteral nutrition within 24 h of intestinal surgery versus later commencement of feeding: a systematic review and meta-analysis. J Gastrointest Surg 2009;13:569-75. [PubMed]
- Osland E, Yunus RM, Khan S, et al. Early versus traditional postoperative feeding in patients undergoing resectional gastrointestinal surgery: a meta-analysis. JPEN J Parenter Enteral Nutr 2011;35:473-87. [PubMed]
- Sekido H, Matsuo K, Takeda K, et al. Impact of early enteral nutrition after liver transplantation for acute hepatic failure: report of four cases. Transplant Proc 2003;35:369-71. [PubMed]
- McClave SA, Martindale RG, Vanek VW, et al. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). JPEN J Parenter Enteral Nutr 2009;33:277-316. [PubMed]
- Marik PE, Zaloga GP. Early enteral nutrition in acutely ill patients: a systematic review. Crit Care Med 2001;29:2264-70. [PubMed]
- Ikegami T, Shirabe K, Yoshiya S, et al. Bacterial sepsis after living donor liver transplantation: the impact of early enteral nutrition. J Am Coll Surg 2012;214:288-95. [PubMed]
- Khalid I, Doshi P, DiGiovine B. Early enteral nutrition and outcomes of critically ill patients treated with vasopressors and mechanical ventilation. Am J Crit Care 2010;19:261-8. [PubMed]
- Wells DL. Provision of enteral nutrition during vasopressor therapy for hemodynamic instability: an evidence-based review. Nutr Clin Pract 2012;27:521-6. [PubMed]
- Braga M, Gianotti L, Gentilini O, et al. Early postoperative enteral nutrition improves gut oxygenation and reduces costs compared with total parenteral nutrition. Crit Care Med 2001;29:242-8. [PubMed]
- Mehta PL, Alaka KJ, Filo RS, et al. Nutrition support following liver transplantation: comparison of jejunal versus parenteral routes. Clin Transplant 1995;9:364-9. [PubMed]
- Wicks C, Somasundaram S, Bjarnason I, et al. Comparison of enteral feeding and total parenteral nutrition after liver transplantation. Lancet 1994;344:837-40. [PubMed]
- Peter JV, Moran JL, Phillips-Hughes J. A metaanalysis of treatment outcomes of early enteral versus early parenteral nutrition in hospitalized patients. Crit Care Med 2005;33:213-20; discussion 260-1. [PubMed]
- Rayes N, Seehofer D, Theruvath T, et al. Supply of pre- and probiotics reduces bacterial infection rates after liver transplantation--a randomized, double-blind trial. Am J Transplant 2005;5:125-30. [PubMed]
- Shirabe K, Yoshimatsu M, Motomura T, et al. Beneficial effects of supplementation with branched-chain amino acids on postoperative bacteremia in living donor liver transplant recipients. Liver Transpl 2011;17:1073-80. [PubMed]
- Yoshida R, Yagi T, Sadamori H, et al. Branched-chain amino acid-enriched nutrients improve nutritional and metabolic abnormalities in the early post-transplant period after living donor liver transplantation. J Hepatobiliary Pancreat Sci 2012;19:438-48. [PubMed]
- Qiu Y, Zhu X, Wang W, et al. Nutrition support with glutamine dipeptide in patients undergoing liver transplantation. Transplant Proc 2009;41:4232-7. [PubMed]
- Zhu XH, Wu YF, Qiu YD, et al. Liver-protecting effects of omega-3 fish oil lipid emulsion in liver transplantation. World J Gastroenterol 2012;18:6141-7. [PubMed]
- Drover JW, Dhaliwal R, Weitzel L, et al. Perioperative use of arginine-supplemented diets: a systematic review of the evidence. J Am Coll Surg 2011;212:385-99. [PubMed]
- Plank LD, McCall JL, Gane EJ, et al. Pre- and postoperative immunonutrition in patients undergoing liver transplantation: a pilot study of safety and efficacy. Clin Nutr 2005;24:288-96. [PubMed]
- Kaido T, Ogura Y, Ogawa K, et al. Effects of post-transplant enteral nutrition with an immunomodulating diet containing hydrolyzed whey peptide after liver transplantation. World J Surg 2012;36:1666-71. [PubMed]
- Reilly J, Mehta R, Teperman L, et al. Nutritional support after liver transplantation: a randomized prospective study. JPEN J Parenter Enteral Nutr 1990;14:386-91. [PubMed]


